Healthcare Barcoding: Why Standard Warehouse Scanners Fail in Clinical Environments
Posted by Midwest Barcoding Solutions on Apr 30th 2026

By Midwest Barcoding Solutions | Healthcare Barcoding | Clinical Device Selection Guide
The healthcare vertical is one of the most demanding environments for barcode hardware for reasons that have nothing to do with ruggedness in the traditional sense. The challenge is not impact resistance or outdoor temperature tolerance. The challenge is infection control, patient safety, regulatory compliance, and the specific physical characteristics of the items being scanned. A wristband on a patient's wrist, a barcode on a glass vial, a medication package wrapped in foil, a specimen label on a curved test tube — these are fundamentally different scanning targets from a warehouse label on a flat cardboard box, and they require a different approach at every level of the hardware stack.
This guide covers the specific ways standard warehouse devices fail in clinical environments, the three regulatory and workflow drivers pushing healthcare facilities toward purpose-built HC-grade hardware, and the devices that MBS carries to address each layer of the healthcare barcoding stack. If you are a healthcare IT director, a clinical informatics manager, or a procurement lead evaluating a device refresh, this is the decision map.
Why Standard Warehouse Scanners Fail in Clinical Environments
The failure modes are specific and documented, not theoretical. Healthcare facilities that have attempted to deploy standard warehouse scanners in clinical settings consistently encounter the same problems.
The Disinfectant Compatibility Problem
Hospital infection control protocols require frequent wiping of all patient-contact surfaces and devices with hospital-grade disinfectants. The active ingredients in these disinfectants include quaternary ammonium compounds, bleach, hydrogen peroxide, isopropyl alcohol, and accelerated hydrogen peroxide formulations. Standard warehouse scanner housings are not formulated to resist these chemicals. The plastics yellow, crack, and become brittle with repeated exposure. Seals around the scan window degrade. Buttons become sticky or unresponsive. Within months of regular hospital cleaning protocols, a standard scanner looks visibly degraded and may no longer function reliably.
Zebra HC-grade devices are built with medical-grade plastics specifically formulated to resist over 30 common hospital disinfectants and cleaning agents. The housing materials are validated against the actual disinfectant products used in hospital environments. This is not a minor material difference. It is the difference between a device that lasts three to five years in clinical use and one that needs replacement within a year.
The Curved Surface Scanning Problem
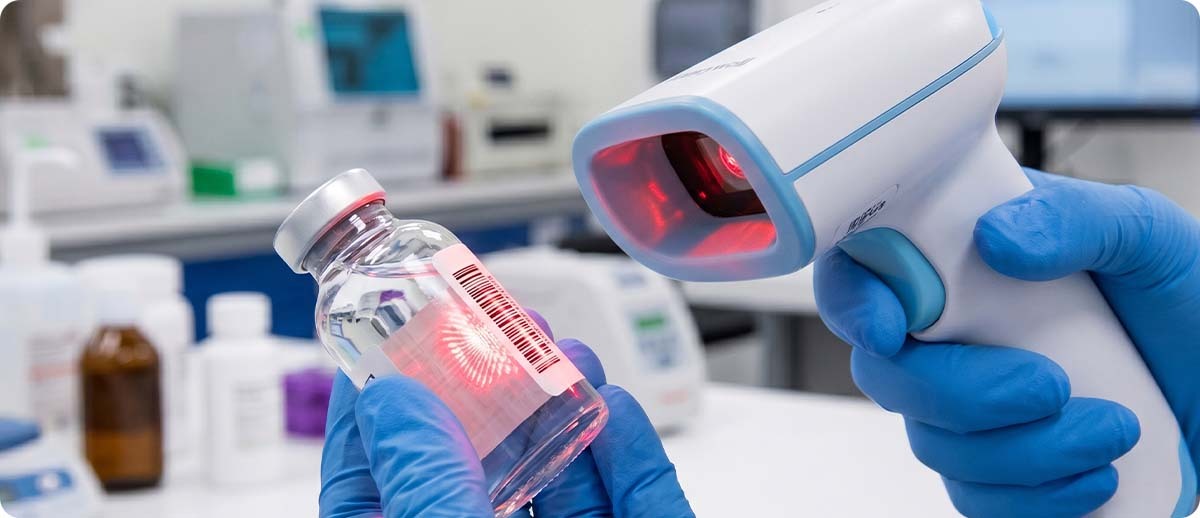
Warehouse scanning is almost entirely flat-surface scanning. Carton labels, pallet labels, shelf labels, and location markers are flat, printed in high contrast, and applied to surfaces that hold still. Clinical scanning involves curved surfaces, reflective packaging, and small or compressed barcodes. IV bags have flexible, curved surfaces that distort the barcode when the bag is full. Syringes have narrow cylindrical barrels where the barcode wraps around the curve. Foil medication packages reflect light in ways that confuse standard scanner optics. Glass vials have curved, often reflective surfaces where a small label carries a dense barcode.
Standard warehouse scanners are optimized for flat, high-contrast, well-lit labels at standard sizes. They consistently fail or produce high first-scan failure rates on curved, reflective, and small-format clinical targets. Healthcare-grade scanners like the Zebra DS8178-HC are specifically engineered and validated to read barcodes on IV bags, syringes, foil medpacks, small vials, and patient wristbands. The scan engine optics, illumination angle, and decode algorithms are tuned for these specific targets, not for warehouse pallet labels.
The Patient Environment Problem
Standard warehouse scanners use a red aiming laser or LED. In a neonatal intensive care unit, the red aiming beam can startle infants, and the audible scan confirmation can disrupt patients in sensitive care environments. Some Zebra HC-series scanner models offer a green aiming LED option that is safer for NICU and other sensitive patient environments, along with configurable audible feedback that can be suppressed or set to a lower volume level appropriate for patient care areas. These are not features a warehouse scanner is designed to offer, because there is no clinical context in a warehouse that requires them.
The Device Design Problem
Warehouse devices are designed to maximize utility in industrial environments. They often have ventilation slots, deep crevices, textured surfaces, and hardware design elements that are appropriate for a warehouse floor but create hygiene problems in clinical settings. Germs and pathogens accumulate in crevices that are difficult to clean. Ventilation slots trap debris. Textured rubber overmolds retain moisture and contaminants after wiping.
HC-grade devices are designed with minimized seams and crevices, covered screw holes, smooth surfaces that can be fully wiped down, and clog-resistant audio grills that resist the entry of cleaning fluids. This hygienic design is engineered from the ground up for clinical use, not retrofitted onto a warehouse device.
The Three Regulatory and Workflow Drivers
1. UDI Compliance: Unique Device Identification
The FDA's Unique Device Identification system requires medical devices to carry a UDI barcode containing device identification information encoded in a standardized format. Class III devices (highest risk) and Class II devices have been subject to UDI requirements since 2014 and 2016 respectively. The UDI barcode must be readable throughout the device's distribution and use lifecycle, and healthcare facilities are expected to scan and record UDI data at points of use and inventory.
UDI barcodes use multiple symbologies including GS1 DataMatrix, GS1-128, and HIBCC formats. They are often printed at very small sizes on curved device packaging. Reading them reliably requires a scanner with a 2D imager capable of reading dense, small-format 2D codes in clinical lighting conditions. Standard warehouse laser scanners do not read 2D codes at all. Even standard 2D warehouse imagers may struggle with the small print sizes and curved surfaces common on medical device packaging. A healthcare-grade scanner validated for UDI reading is the correct specification for any facility capturing UDI data at the point of use.
2. Medication Administration Verification (BCMA)
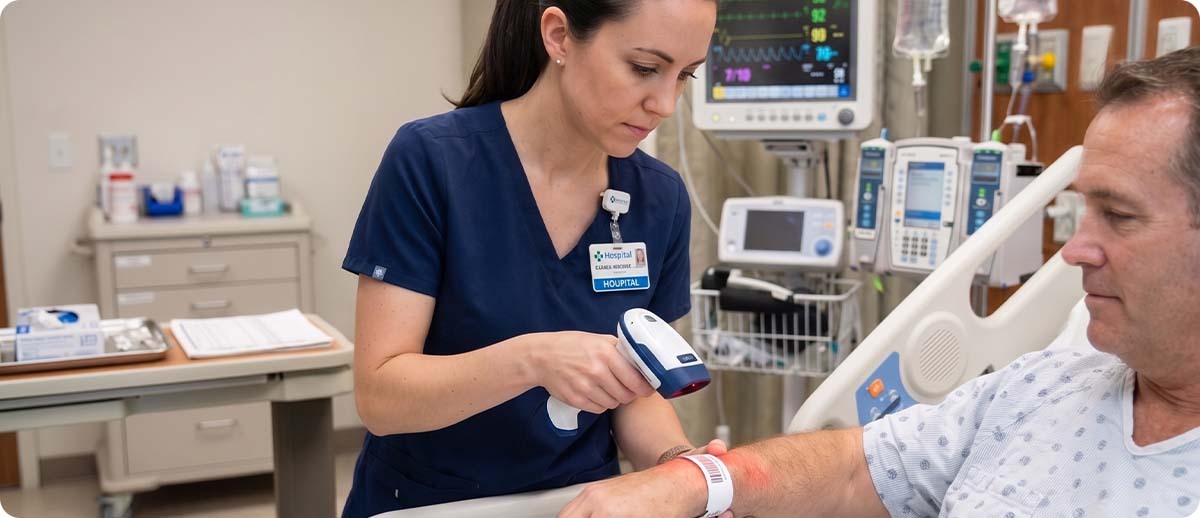
Barcode Medication Administration systems require nurses and pharmacists to scan the patient wristband and the medication barcode at the point of care before administration, verifying the Five Rights of medication administration: right patient, right drug, right dose, right route, right time. BCMA systems have documented reductions in medication errors of 50 to 80 percent in facilities that implement them correctly.
BCMA workflow places specific demands on the scanner hardware. The scanner must reliably read patient wristband barcodes, which may be smeared, creased, or partially obscured after hours of wear. It must read medication package barcodes, which are often small, on curved blister packaging or foil materials, in poor lighting at the bedside. Scan failures in a BCMA workflow are not just productivity problems; they create clinical risk. A nurse who cannot scan a medication after multiple attempts may override the system, introducing exactly the verification gap the system is designed to prevent. Scanner reliability in BCMA is a patient safety issue, not just an operational one.
3. Patient Identification and Wristband Printing
Accurate patient identification underpins every clinical workflow. The Joint Commission identifies patient misidentification as a leading cause of sentinel events, and wristband-based patient ID using barcodes is the standard mechanism for addressing it. The wristband barcode must print correctly, remain readable throughout the patient's stay, and scan reliably on the patient's wrist in conditions that include water, movement, and curved surfaces.
This requires a wristband printer, not a standard label printer. Zebra's wristband media is printed on materials specifically designed for patient skin contact, antimicrobial-coated, water-resistant, and formulated to maintain barcode readability over extended wear periods. Standard label stock is not appropriate for direct patient contact or for the wear and moisture exposure of a patient wristband. The ZD510-HC, Zebra's dedicated wristband printer, uses Z-Band cartridges containing Zebra's antimicrobial-coated wristband media and is specifically designed for bedside wristband printing in clinical environments.

The Healthcare Hardware Stack at MBS
A complete clinical barcoding deployment covers three hardware layers: point-of-care scanners for medication and patient ID verification, mobile computers for nurses and pharmacists who need a full computing platform at the bedside, and wristband printers for patient identification at admissions and on nursing floors. MBS carries confirmed HC-grade products at each layer.
Layer 1: Point-of-Care Scanners
Zebra DS8178-HC Healthcare Cordless Scanner
Part #: DS8178-HCBU210MP5W (USB kit, standard) | DS8178-HCMF00BVMWW (scanner only) | DS8178-HCBU210FS5W (USB kit, green LED for NICU)
The DS8178-HC is Zebra's flagship cordless healthcare scanner, purpose-built and validated for clinical workflows. It reads barcodes on IV bags, foil medpacks, syringes, small vials, and patient wristbands in conditions where standard healthcare scanners struggle. The housing withstands frequent cleaning with the widest range of hospital disinfectants. Multiple feedback modes including audible, visual, and tactile allow configuration for specific care environments. The green LED aiming version (DS8178-HCBU210FS5W) is the correct specification for NICU and other light-sensitive patient care areas where the standard red aimer would be clinically inappropriate. Cordless operation with a healthcare-grade cradle allows placement at the bedside or medication cart without cable management concerns.
Shop Zebra DS8178-HC →Zebra DS8108-HC Healthcare Corded Scanner
Part #: DS8108-HCBU2104ZVW (USB kit)
The DS8108-HC brings the same clinical scanning capability as the DS8178-HC in a corded configuration for applications where a tethered scanner is preferred: pharmacy dispensing stations, nursing workstations on wheels, and admissions desks where the scanner is stationary and battery management is not a factor. Same disinfectant-ready plastics, same validated scanning performance on IV bags, medpacks, syringes, and vials. The corded design eliminates battery tracking and charging management, which simplifies IT management in high-traffic pharmacy and nursing station environments.
Shop Zebra DS8108-HC →Datalogic Gryphon GM4500-HC Healthcare Scanner
Part #: GM4500-HC-433K1 (USB kit, 433 MHz cordless)
Datalogic's Gryphon HC series provides an alternative healthcare scanner platform with antimicrobial additives built directly into the housing material, providing ongoing surface microbial suppression between cleaning cycles. Reads 1D and 2D barcodes including codes from LCD monitors, smartphones, and mobile devices, which supports digital prescription verification and EHR display scanning workflows. Compatible with the WLC4090-HC-433 healthcare cradle included in the kit configuration. For facilities standardized on Datalogic hardware or requiring an antimicrobial-additive housing in addition to disinfectant-ready materials, the GM4500-HC is a strong clinical scanner option.
Shop Datalogic GM4500-HC →Layer 2: Healthcare Mobile Computers
Point-of-care workflows that require a nurse or pharmacist to access the EHR, review patient records, document medication administration, and scan barcodes in a single workflow need a mobile computing platform, not just a scanner. The scanner is the data capture element. The mobile computer is the clinical application platform.
HC-grade mobile computers carry all the same disinfectant-resistant design requirements as HC scanners, plus additional clinical features: an emergency alarm button for worker safety in patient care areas, three-microphone configurations for noise cancellation during clinical communications, high-volume speakers for clear alerting, push-to-talk clinical communication integration, and NFC for proximity-based patient ID and medication verification in workflows where card-tap is part of the authentication process.
MBS carries healthcare mobile computers including the Zebra TC52ax-HC, which builds on the highly successful TC52-HC with Wi-Fi 6 support, double the memory of the original TC52-HC, healthcare blue colors designed with materials supporting over 30 chemical disinfectants and UV-C cleaning appliances, and a BLE beacon battery that enables device location tracking through an EMM without additional hardware. The TC52ax-HC is the current-generation clinical mobile computing platform for bedside medication administration, patient identification, and clinical documentation workflows.
View Healthcare Mobile Computers at MBS →
Layer 3: Wristband Printing
Zebra ZD510-HC Healthcare Wristband Printer
Part #: ZD51013-D01E00FZ
The ZD510-HC is Zebra's dedicated patient wristband printer, combining a disinfectant-ready UV-resistant housing with cartridge-loaded Z-Band wristband media. Z-Band wristbands are the only antimicrobial-coated patient wristbands on the market, designed for skin contact, water resistance, and barcode readability through the full duration of a patient stay. More than 382.8 million patients globally have been identified using Zebra patient wristbands. The cartridge system makes wristband loading error-resistant at the bedside, where nursing staff should not be troubleshooting media loading under shift pressure. Link-OS connectivity for integration with EMR-triggered wristband printing from admission systems. For facilities still printing wristbands on desktop label printers with standard label media, the ZD510-HC and Z-Band system is a meaningful quality and safety upgrade.
Shop Zebra ZD510-HC →Zebra ZD421-HC Healthcare Desktop Printer
Part #: ZD4AH42-D01E00EZ
The ZD421-HC is Zebra's healthcare-grade desktop label printer for clinical environments that need to print beyond wristbands: specimen labels, pharmacy labels, asset tags for medical equipment, and laboratory sample labels. The ZD421-HC carries the same disinfectant-resistant housing as HC mobile devices, validated for hospital cleaning protocols. 30 percent more powerful than the previous ZD420 series with a field-upgradeable wireless kit for connectivity flexibility. Available in direct thermal configuration for short-duration clinical labels. Link-OS for centralized print management across a hospital's printer fleet. The correct specification when a healthcare facility needs a general-purpose clinical label printer that can be cleaned to hospital infection control standards.
Shop Zebra ZD421-HC →
HC vs. Standard: The Specification Comparison
| Requirement | Standard Warehouse Device | Zebra HC-Grade Device |
|---|---|---|
| Hospital disinfectant compatibility | Not validated; housing degrades | Validated for 30+ disinfectants |
| Curved surface scanning (vials, syringes, IV bags) | High failure rate on curved/reflective targets | Specifically validated for clinical targets |
| NICU / sensitive environment aimer | Red aimer only; not configurable | Green LED option; configurable audio |
| Hygienic housing design | Crevices, vents, textured surfaces | Minimized seams, covered screws, smooth surfaces |
| 2D barcode reading (UDI, BCMA) | Available but not optimized for clinical use | Validated for UDI and BCMA symbologies |
| Patient wristband printing | Not designed for wristband media or patient contact | ZD510-HC with Z-Band antimicrobial wristbands |
| HIPAA / data security | Standard enterprise security | Healthcare-grade security with HIPAA compliance tools |
Frequently Asked Questions: Healthcare Barcoding
Can we just use any 2D barcode scanner for UDI compliance or does it have to be healthcare-specific?
Any 2D imager that can read GS1 DataMatrix and GS1-128 symbologies can capture UDI data at a technical level. The regulatory requirement is data capture accuracy, not the device type. However, the practical performance difference in clinical environments is significant. Standard 2D warehouse imagers are not validated for the curved, reflective, and small-format targets common on medical device packaging. A scanner that reads a flat warehouse label reliably may produce unacceptably high failure rates on a glass vial or a foil blister pack. For any facility capturing UDI data at the point of use, an HC-grade scanner validated for clinical scanning targets is the appropriate specification for reliable compliance workflow performance.
What disinfectants specifically are HC-grade Zebra devices validated for?
Zebra publishes a disinfectant compatibility list for each HC device. The validated disinfectants include quaternary ammonium compounds such as Sani-Cloth AF3 and Sani-Cloth Plus, bleach-based solutions, hydrogen peroxide formulations, isopropyl alcohol, and accelerated hydrogen peroxide products such as Oxivir. The full compatibility list varies by specific device model. Before deploying any HC device in a facility with specific infection control protocols, it is worth verifying that the device's validated disinfectant list includes the specific products used in that facility's cleaning program. Zebra's cleaned and disinfected guidelines are available for each HC device model, and MBS can provide specific documentation on request.
We currently print patient wristbands on a standard desktop label printer. What is the problem with that?
Standard desktop label printers and standard label stock are not designed for patient contact or for the conditions a wristband experiences during a hospital stay. Standard label adhesive and facestock are not formulated for skin contact, water resistance, or repeated flexing on a patient's wrist. The barcode on a standard label may degrade within hours of application, particularly on pediatric patients or in environments with frequent hand washing. Zebra Z-Band media is specifically engineered for wristband applications with antimicrobial coating, water resistance, and barcode durability through extended patient wear. The ZD510-HC printer and Z-Band cartridge system is Zebra's validated solution for this application, and the clinical risk difference between it and a standard desktop printer with standard label stock is not insignificant.
Are HC mobile computers backward compatible with standard Zebra accessories?
It depends on the specific device generation. The TC52ax-HC is backward compatible with TC52-HC accessories including charging cradles, holsters, cases, and most carrying solutions. This is a meaningful cost consideration for facilities upgrading from TC52-HC to TC52ax-HC, as existing accessory infrastructure carries forward. For facilities upgrading from older generations such as TC51-HC, accessory compatibility requires verification by device. MBS can provide an accessory compatibility review as part of a device refresh evaluation.
Healthcare barcoding is a different technical conversation from warehouse barcoding, and getting the hardware specification wrong has consequences that go beyond operational inefficiency. If you are evaluating a clinical device refresh, building out a BCMA program, or simply trying to understand what your current standard devices cannot do in a clinical environment, our team has worked through this evaluation with healthcare facilities across the region. Fill out the form below and let's make sure your hardware matches what your clinical environment actually requires.
